In the production of pharmaceuticals, patient safety must come first.
Pharmaceutical & Biopharmaceutical Industry Products
Overview
Through developments in drug discovery, the industry is finding new, innovative ways to treat and even prevent diseases. Not only does the pace of research and discovery continue to quicken, with these new possibilities for patient well-being comes greater need for security: To ensure pharmaceutical products are safe for patients, they must be manufactured and packaged to minimize risk to the user as required by stringent regulations.
Gore helps those in both drug discovery and manufacturing compete in this increasingly regulated landscape with reliable solutions for purifying, transferring, storing, filtering, processing and packaging. These solutions include durable, multi-use process components and single-use products that enable researchers and manufacturers to control costs through process improvements — highly important given the escalating costs of biologics development.
Backed by our materials science expertise and commitment to regulatory compliance, we have a track record of improving the processing and delivery of pharmaceutical products.
Meeting the Industry’s Changing Needs
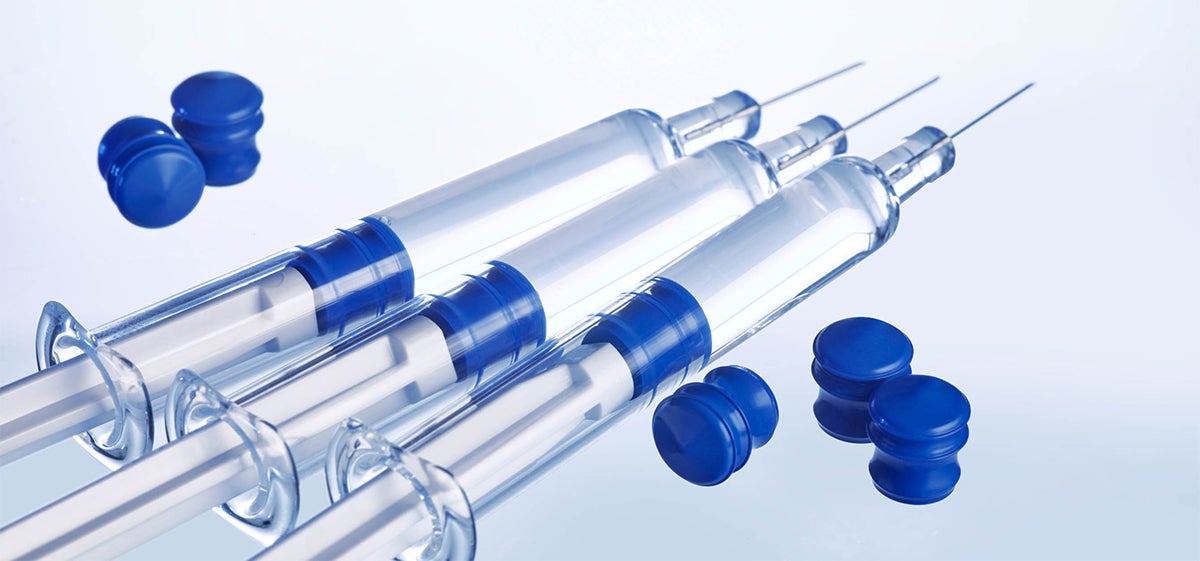
Eliminate silicone in both barrel and plunger to protect sensitive biologics in PFS.
Today's "breakthrough" biologic drug products may require packaging technology that is equally innovative – especially when these high-value, complex formulations are silicone-sensitive. Our newest solution, the GORE® IMPROJECT® Plunger for pre-filled syringes (PFS), is designed for use in bare glass (non-siliconized) barrels, and the plunger itself is silicone-free – silicone oil is neither a raw material, nor used in manufacturing of the plungers. By eliminating the need for silicone as a lubricant on the plunger and in the barrel, these plungers can help to avoid silicone-induced aggregation and particulation, while enabling consistent delivery performance with time. And, due to its proprietary plunger design and fluoropolymer barrier, the GORE IMPROJECT Plunger ensures no significant extractable compounds.
In recent decades, Gore has focused its expertise in fluoropolymers on developing new, technology-driven solutions for the life sciences, particularly for pharmaceutical and biopharmaceutical applications.
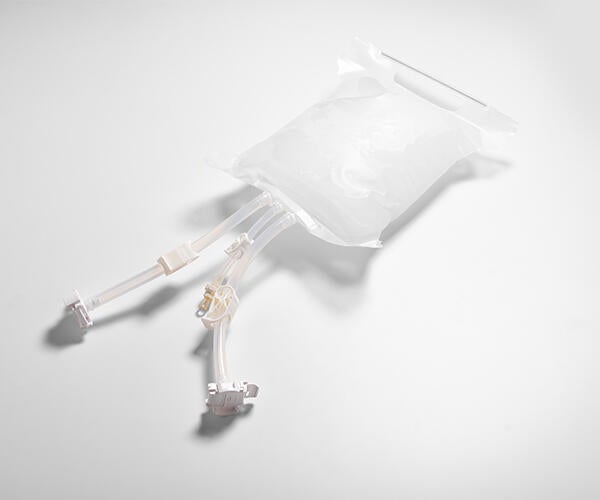
In the early 2000’s, we saw an opportunity to contribute to the pharmaceutical industry with the release of the GORE® LYOGUARD® Freeze-Drying Tray.

The breathability of ePTFE gives GORE LYOGUARD Freeze-Drying Trays its superior vapor transmission rate.
The single-use tray incorporates an ePTFE membrane selected for its superior barrier properties and high vapor transmission rate. The tray’s high vaporization rate allows materials to freeze-dry very quickly — while its barrier protection minimizes the risk of operator exposure to APIs and helps reduce or eliminate cleaning and validation costs.
Over time, we’ve added more transfer and containment solutions to our portfolio, leveraging our materials science expertise and additional ePTFE benefits, like its biocompatibility, high tensile strength and low particulation.
Single-use GORE® STA-PURE Flexible Freeze Containers offer a new solution for bulk drug substance that protects container integrity with greater crack- and impact-resistance during cold chain handling. The high-purity fluoropolymer container material has a low extractables profile for added piece of mind.

GORE STA-PURE Pump Tubing performs reliably under challenging processing conditions.
GORE® STA-PURE® Pump Tubing provides durable and reliable product performance in long runs and under high pressure or other challenging process conditions. Our other single-use components, such as GORE Molded Assemblies, enable the secure transfer, containment and sampling of liquids during pharmaceutical manufacture.
As the industry sought quality and process improvements, Gore leveraged its decades of experience in filtration and venting to serve pharmaceutical and biopharmaceutical manufacturers. Now, we offer several proven products for filtration:
GORE® Microfiltration Media is readily incorporated into filter devices. Our various forms of filter media offer effective and reliable retention of particles to provide superior efficiency and process security.
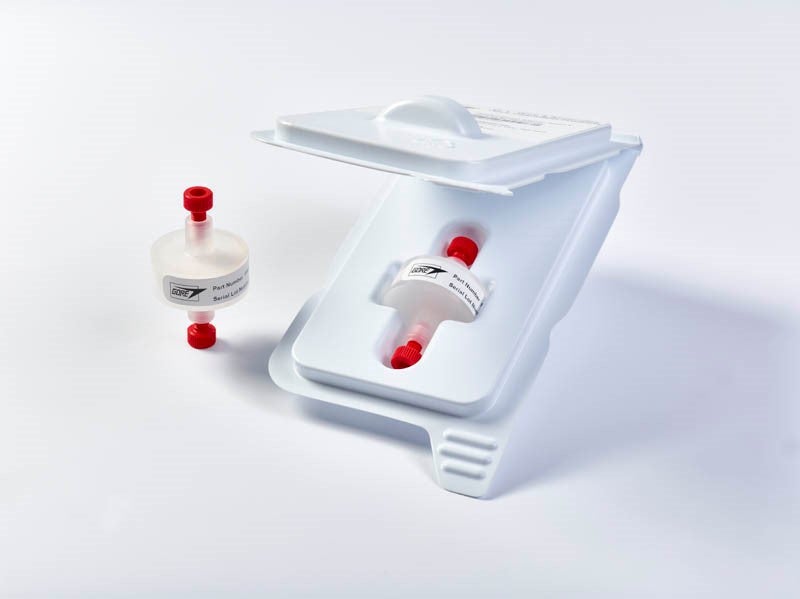
GORE® Protein Capture Devices are ideal for drug discovery applications, providing high binding capacity and short residence times.
Researchers in biologic drug development depend on repeatable and reliable processes to deliver high quality results. Traditional protein purification processes can be slow and often require additional steps to achieve needed concentration.
The GORE® Protein Capture Device with Protein A utilizes a unique membrane solution for faster antibody purification to help minimize screening pipeline back-ups, help eliminate downstream concentration steps, and improve the speed of purification.
Products that Perform as Promised
With Gore’s materials science expertise and collaboration with the pharmaceutical industry comes an understanding of how our solutions provide value in a given application.
When developing a product or process component, we tailor our materials and performance targets to suit the customer’s system requirements, often going beyond what’s expected. Prior to releasing the product, we rigorously test it for application-readiness to ensure peak performance and reliability over time.
Good Manufacturing Processes
All Gore products for life sciences are manufactured specifically for use in medical or pharmaceutical settings and comply with regulations our customers may require. Depending on those requirements, you will find products manufactured under Good Manufacturing Practices (GMP), ISO 13485, ISO 15378, and/or regulations for food contact.
Contact Us
Europe
+49 89 4612 3456
+800 4612 3456
Americas
+1 800 294 4673
+1 410 506 1715
中国
+86 21 5172 8299
+86 21 6247 9199 (传真)
Southeast Asia
+65 6210 6946
Japan
+81 3 6746 2570
US
+1 800 294 4673
+1 410 506 1715
Resources

Brochure: Trusted Solutions for Medical Device Manufacturers
Brochures, 15 MB

Validation Guide: GORE® STA‑PURE® Pump Tubing, Series PCS
Validation Guides

Handling Guide: GORE® LYOGUARD® Freeze-Drying Trays
Technical Information, 132.42 KB